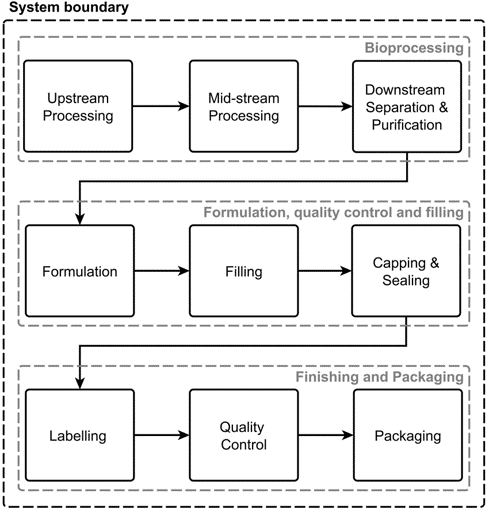
AI plays a crucial role in anomaly detection, predictive maintenance, and enhancing quality testing processes.
Human errors often lead to low success rates.
AI can significantly reduce processing time and enhance speed through anomaly detection, resulting in fewer failed batches. It can also expedite quality control procedures, reducing long lead times. Additionally, AI-powered data fusion aids in swift regulatory filings.
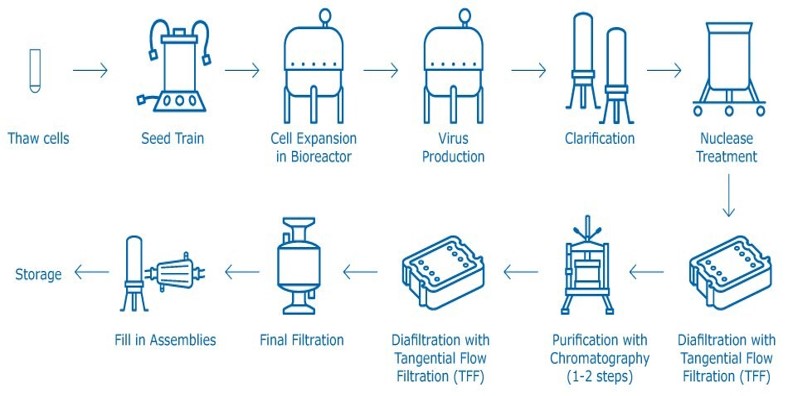
AI-ML🡺 higher yields, Less batch Loss
Time Saved: Could be 3-6 months
AI-ML based Quality Control:
Typical lead time is 4 months, The AI-ML can reduce the time needed to 1 month and higher accuracy